From the Open-Publishing Calendar
From the Open-Publishing Newswire
Indybay Feature
Prop 64 Has Passed: Now What? "Track and Trace" May Become Widespread
Swiss-based firm SICPA Hoping to Expand Successful Humboldt Program Statewide
Now that Prop 64 has passed, many Californians are wondering: what’s next? How will the state manage compliance requirements in the cannabis industry, safeguard public health, address public safety concerns, ensure proof of origin and manage taxation?
The answer may be a medical cannabis control solution pilot that Humboldt County is running in partnership with SICPA, a global leader in security solutions. In its first two months, this county program has helped regulate 970 pounds of medical cannabis working across the supply chain from cultivator to patient. This solution was designed with the potential for recreational cannabis legalization and can serve as the model for the rest of the state.
The program is based on SICPA’s proven track and trace solution, SICPATRACE®, which combines counterfeit-resistant stamps, traceability and enforcement tools, and data management services to create a platform that ensures easy regulatory compliance. This solution documents a forensic trail across the medical cannabis supply chain, from cultivator to dispensary, enabling government officials and law enforcement to accurately track products, identify and reduce the spread of illegal cannabis, and bring criminals to justice.
With Prop 64, taxation of cannabis will become a reality. SICPA’s solution can easily support a tax component. In fact, the California Board of Equalization has used SICPATRACE® since 2005 to effectively track and trace approximately 900 million packs of cigarettes annually, representing more than $835 million in annual excise tax collections.
How the Medical Cannabis Control Solution Works
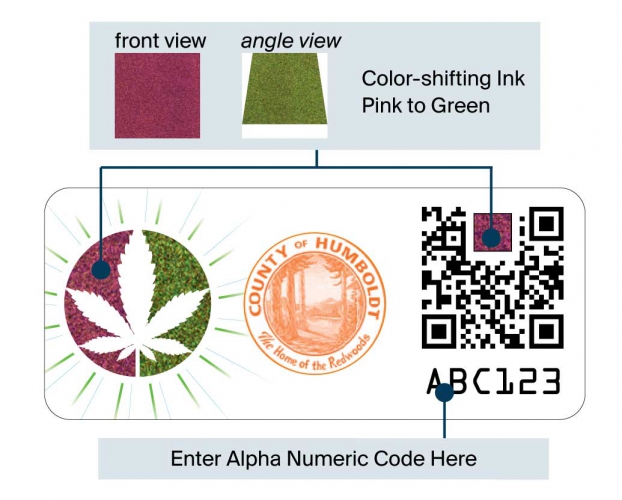
Cultivators document their product catalog (e.g., strain, growing techniques, THC content) and apply traceable, counterfeit-resistant stamps to product packaging. Each stamp includes a secure QR code and unique number that is encoded with details from the product catalog, package size, and transfer details. As the package moves through the supply chain—from cultivator to manufacturer to distributor to dispensary—the stamps are coded with more information and create a map of a product’s journey through the system.
The stamps will also connect patients to product catalog information through a smartphone app and website. Patients can use these tools to make sure a product is authentic, retrieve product details, see test results, and more.
The answer may be a medical cannabis control solution pilot that Humboldt County is running in partnership with SICPA, a global leader in security solutions. In its first two months, this county program has helped regulate 970 pounds of medical cannabis working across the supply chain from cultivator to patient. This solution was designed with the potential for recreational cannabis legalization and can serve as the model for the rest of the state.
The program is based on SICPA’s proven track and trace solution, SICPATRACE®, which combines counterfeit-resistant stamps, traceability and enforcement tools, and data management services to create a platform that ensures easy regulatory compliance. This solution documents a forensic trail across the medical cannabis supply chain, from cultivator to dispensary, enabling government officials and law enforcement to accurately track products, identify and reduce the spread of illegal cannabis, and bring criminals to justice.
With Prop 64, taxation of cannabis will become a reality. SICPA’s solution can easily support a tax component. In fact, the California Board of Equalization has used SICPATRACE® since 2005 to effectively track and trace approximately 900 million packs of cigarettes annually, representing more than $835 million in annual excise tax collections.
How the Medical Cannabis Control Solution Works
Cultivators document their product catalog (e.g., strain, growing techniques, THC content) and apply traceable, counterfeit-resistant stamps to product packaging. Each stamp includes a secure QR code and unique number that is encoded with details from the product catalog, package size, and transfer details. As the package moves through the supply chain—from cultivator to manufacturer to distributor to dispensary—the stamps are coded with more information and create a map of a product’s journey through the system.
The stamps will also connect patients to product catalog information through a smartphone app and website. Patients can use these tools to make sure a product is authentic, retrieve product details, see test results, and more.
Add Your Comments
We are 100% volunteer and depend on your participation to sustain our efforts!
Get Involved
If you'd like to help with maintaining or developing the website, contact us.
Publish
Publish your stories and upcoming events on Indybay.
Topics
More
Search Indybay's Archives
Advanced Search
►
▼
IMC Network